Explore the AP Chemistry Lab Manual
The AP Chemistry Lab Manual, provided by the College Board, serves as an invaluable resource for both educators and students participating in the AP Chemistry program. https://apcentral.collegeboard.org/courses/ap-chemistry/classroom-resources/lab-manual The AP Chemistry Lab Manual comprises 16 student-directed guided-inquiry labs. Their purpose is to provide students with opportunities to engage in hands-on experiments and develop essential laboratory skills, which can greatly enhance their understanding of the course material and prepare them for the AP Chemistry exam. Navigating the AP Chemistry course is undeniably intensive, characterized by an extensive content load, often compounded by students lacking essential prerequisite skills, and exacerbated by tight time constraints. In such an environment, the incorporation of labs, which are inherently time-consuming, can pose a significant challenge. Here at Viziscience, we understand the challenges that teachers face in preparing their students for success in such a demanding course. That’s why we’ve taken a unique approach, one that combines both theoretical understanding and hands-on practicals. Our labs are thoughtfully designed not only to bolster students’ theoretical comprehension but also to provide them with hands-on experience. In doing so, we aim to equip both students and teachers with a comprehensive toolset that enables them to navigate the demanding landscape of AP Chemistry successfully.
Viziscience’s AP Chemistry Online Lab:
Exploring Differences Between Ionic and Covalent Compounds
Objective:
The primary objective of this online lab is to investigate the fundamental differences between ionic and covalent compounds in a manner that aligns with AP Chemistry curriculum, emphasizing both course content and scientific practices. Through this lab, students will:
Aligning with AP Chemistry:
1. Course Content Emphasis:
Our lab meticulously integrates with AP Chemistry course content in the following ways:
- Understanding Melting/Boiling Points: Students will delve into the relationship between chemical structure and melting/boiling points, gaining insights into the distinctive characteristics of ionic and covalent compounds. This aligns with the AP Chemistry learning objectives related to chemical properties.
- Electrolytic Properties: The exploration of solubility and dissociation behavior is a gateway to comprehending electrolytic properties—a key topic in AP Chemistry. By examining how compounds conduct electricity, students hone their ability to differentiate between ionic and covalent compounds, linking directly to course objectives.
2. Science Practices Integration:
Our labs seamlessly integrate scientific practices, cultivating essential skills needed for success in AP Chemistry and scientific inquiry in general:
- Planning and Carrying Out Investigations: Students follow systematic procedures, enabling them to conduct controlled experiments. This aligns with the AP Chemistry skill of designing experiments.
- Analyzing and Interpreting Data: Through data collection and analysis, students sharpen their ability to draw meaningful conclusions—a skill central to the AP Chemistry curriculum.
- Engaging in Scientific Reasoning: By applying chemical concepts to explain observations, students develop critical thinking skills. These skills are vital for addressing complex chemistry problems, in line with AP Chemistry expectations.
- Communicating Findings: Effective communication of results, whether through written explanations or graphical representations, fosters skills essential for the AP Chemistry exam and scientific discourse.
1. Course Content Alignment:
a. Understand Melting/Boiling Points:
- Explore the relationship between the chemical structure of compounds and their melting and boiling points.
- Identify and classify compounds as either ionic or covalent based on their properties.
- Examine the impact of intermolecular forces on the physical state of compounds at room temperature.
b. Electrolytic Properties:
- Investigate the solubility and dissociation behavior of compounds in water.
- Recognize the difference between electrolytes and non-electrolytes and their conductivity properties.
- Identify specific covalent compounds that can dissociate into ions in aqueous solutions, such as acids and bases.
2. Science Practices Alignment:
a. Planning and Carrying Out Investigations:
- Develop and implement a systematic procedure to measure and compare the melting points of selected compounds.
- Conduct controlled experiments to test the electrical conductivity of compounds in solution.
- Collect, record, and analyze data to draw conclusions about the nature of the compounds.
b. Analyzing and Interpreting Data:
- Interpret the experimental results to differentiate between ionic and covalent compounds based on their properties.
- Draw conclusions about the behavior of compounds in different states and their ability to conduct electricity.
c. Engaging in Scientific Reasoning:
- Apply chemical concepts, such as ionic bonding, covalent bonding, intermolecular forces, and dissociation, to explain the observed phenomena.
- Analyze the relationship between chemical structure and physical properties.
d. Communicating Findings:
- Communicate experimental results and conclusions effectively through written explanations, data tables, and graphical representations.
Leveraging Time and Resources:
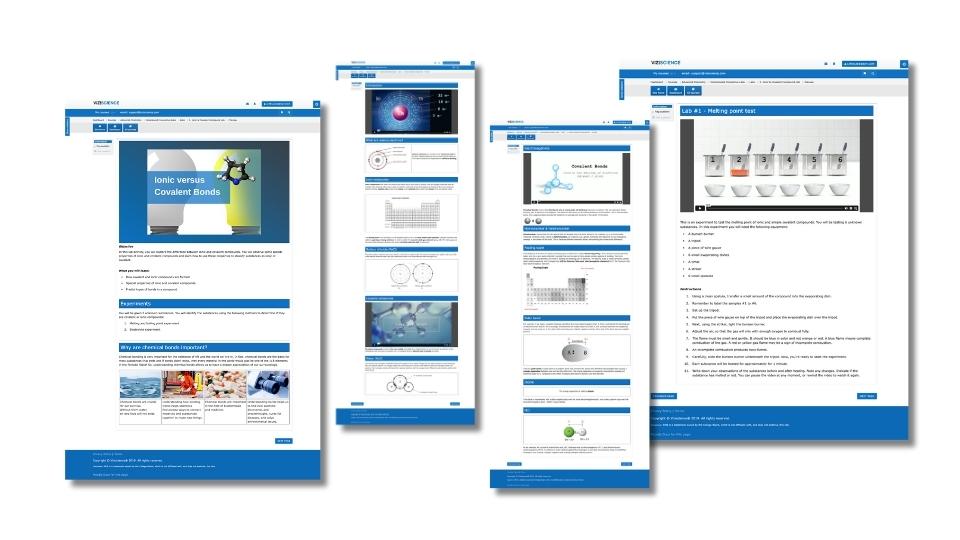
Hands-on Experiments for Success in AP Chemistry
One of the distinctive advantages of our lab approach is efficiency. Our labs are designed with accuracy in materials and quantities, allowing for repeatability in wet labs. Teachers can capitalize on this advantage by using our labs as:
- Pre-Labs: As an essential preparatory step before wet labs, our online labs provide students with a hands-on preview of concepts and techniques. This not only saves valuable classroom time but also enhances the learning experience in the wet lab.
- Time Constraint Mitigation: In a fast-paced AP Chemistry course, time is a precious commodity. Our labs offer a solution by condensing essential learning into an efficient online format, perfect for addressing time constraints.
- Review and Post-Lab: Our labs are versatile tools for reinforcing and reviewing concepts. After students have completed wet labs, our online labs provide a platform for review, consolidation, and revision.
By engaging with our labs, students gain a solid foundation in the topic of ionic and covalent bonding properties. These labs enable teachers to achieve a dual educational purpose: efficient and effective preparation for AP Chemistry and a robust understanding of the principles underpinning ionic and covalent compounds.


Your blog is a true hidden gem on the internet. Your thoughtful analysis and engaging writing style set you apart from the crowd. Keep up the excellent work!